 What is an electron?Įlectrons are tiny compared to protons and neutrons, over 1,800 times smaller than either a proton or a neutron. 
Three quarks make up each proton - two "up" quarks (each with a two-thirds positive charge) and one "down" quark (with a one-third negative charge) - and they are held together by other subatomic particles called gluons, which are massless. Elements are arranged in the Periodic Table of the Elements in order of increasing atomic number. The number of protons also determines the chemical behavior of the element. The number of protons in an atom is referred to as the atomic number of that element. For example, carbon atoms have six protons, hydrogen atoms have one and oxygen atoms have eight. The number of protons in an atom is unique to each element. Protons are about 99.86% as massive as neutrons according to the Jefferson Lab. Rutherford discovered them in experiments with cathode-ray tubes that were conducted between 19. Protons are positively charged particles found within atomic nuclei. These atoms will then decay into other elements, such as carbon-14 decaying into nitrogen-14. Some atomic nuclei are unstable because the binding force varies for different atoms based on the size of the nucleus. This force between the protons and neutrons overcomes the repulsive electrical force that would otherwise push the protons apart, according to the rules of electricity. The nucleus is held together by the strong force, one of the four basic forces in nature. The protons and neutrons that make up the nucleus are approximately the same mass (the proton is slightly less) and have the same angular momentum, or spin. Virtually all the mass of an atom resides in its nucleus, according to Chemistry LibreTexts. He also theorized that there was a neutral particle within the nucleus, which James Chadwick, a British physicist and student of Rutherford's, was able to confirm in 1932. In 1920, Rutherford proposed the name proton for the positively charged particles of the atom. The nucleus was discovered in 1911 by Ernest Rutherford, a physicist from New Zealand, according to the American Institute of Physics. Adding a proton to an atom makes a new element, while adding a neutron makes an isotope, or heavier version, of that atom. Atoms always have an equal number of protons and electrons, and the number of protons and neutrons is usually the same as well. However, one proton is about 1,835 times more massive than an electron. “The more analyses we perform, the more kinds of exotic hadrons we find,” physicist Niels Tuning said in a statement.Protons and neutrons have approximately the same mass. Scientists at CERN say they have observed a new kind of “pentaquark” and the first-ever pair of “tetraquarks” - adding three members to the list of new hadrons. More rarely, however, they can also combine into four-quark and five-quark particles, or tetraquarks and pentaquarks. Quarks are elementary particles that usually combine in groups of twos and threes to form hadrons such as the protons and neutrons that make up atomic nuclei. They will help physicists better understand how quarks bind together into composite particles.  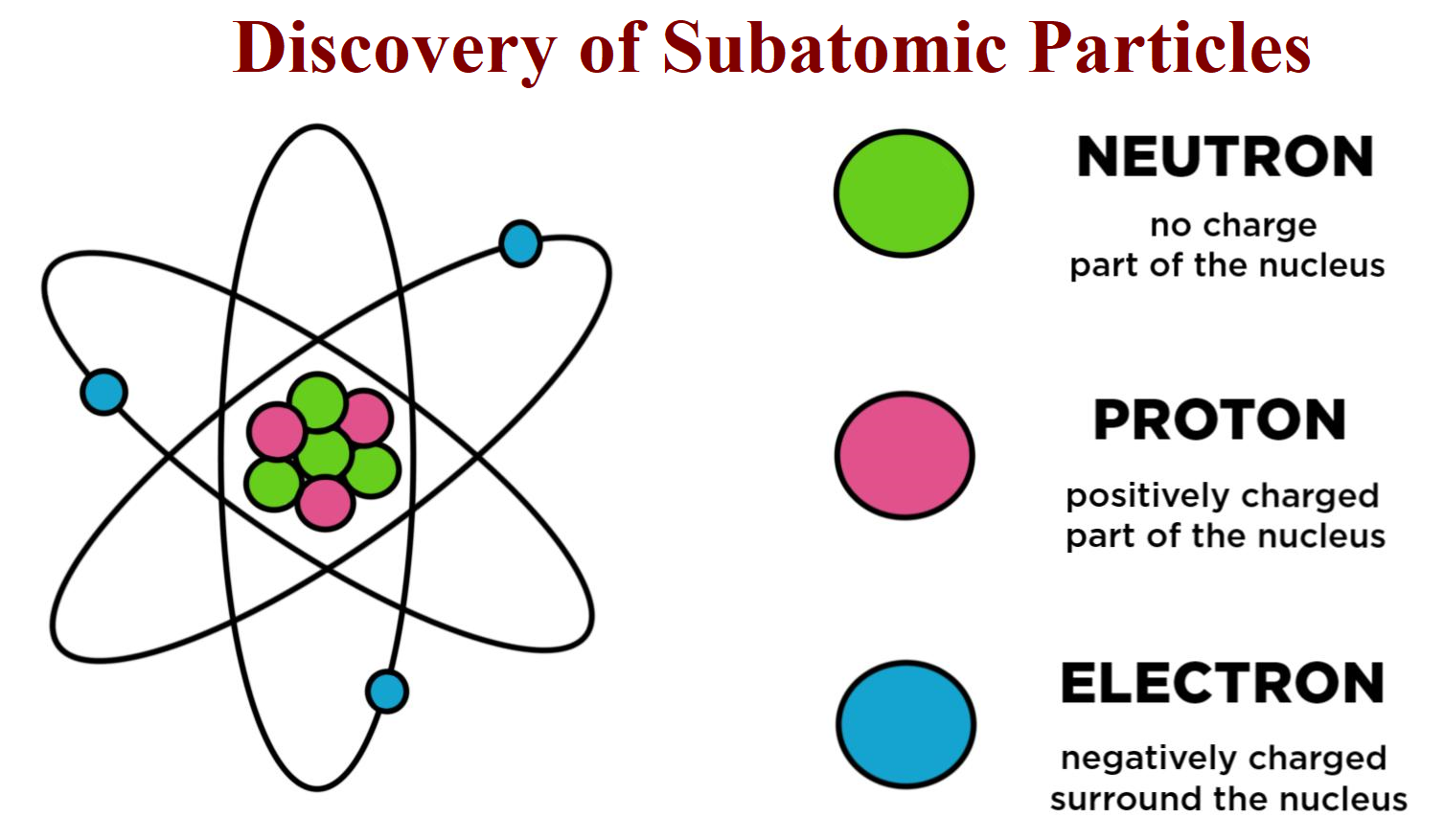
Now scientists at CERN say they have observed a new kind of “pentaquark” and the first-ever pair of “tetraquarks,” adding three members to the list of new hadrons found at the LHC. 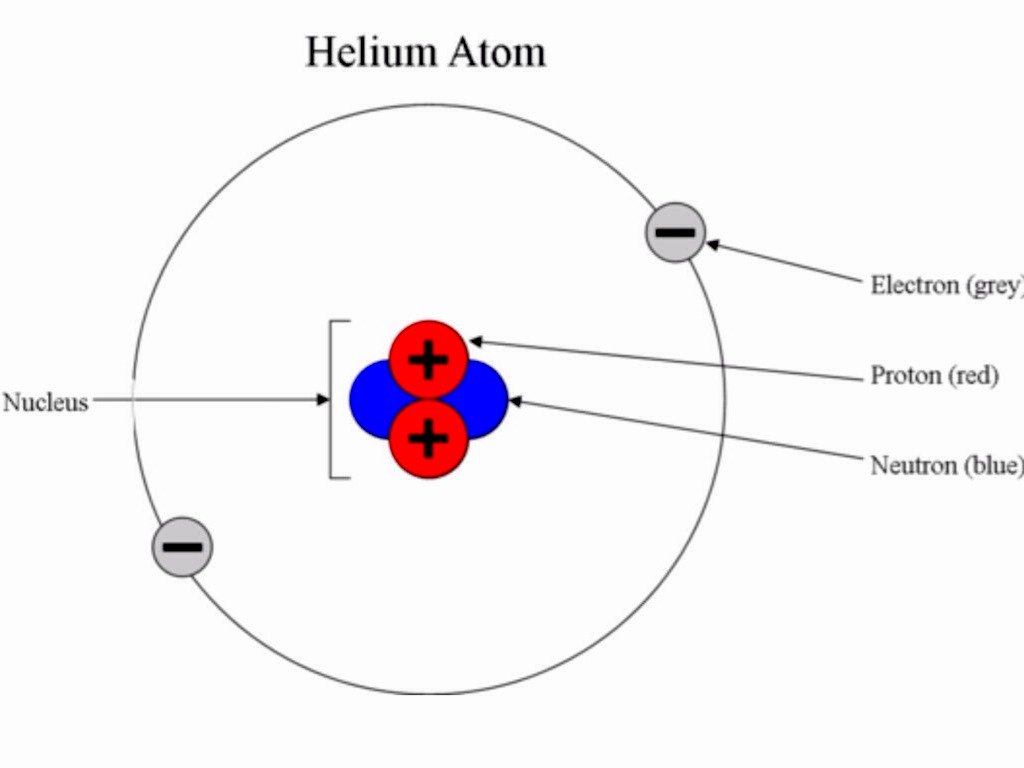
The 16.8 mile-long LHC at CERN is the machine that found the Higgs boson particle, which along with its linked energy field is thought to be vital to the formation of the universe after the Big Bang 13.7 billion years ago. Scientists working with the Large Hadron Collider have discovered three subatomic particles never seen before as they work to unlock the building blocks of the universe, the European nuclear research center CERN said on Tuesday. This superfood can protect your gut and ward off disease: new studyĪI is more than ChatGPT - is the sci-fi cliché about to turn real? ‘Hidden’ Bible chapter rediscovered 1,750 years later with UV photography MIT scientists discover ‘remarkable’ way to reverse Alzheimer’s disease 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
